|
Now that we know what sigma and pi bonds are and when they appear in single, double, and triple bonds, the only thing left is to put our knowledge into action! Counting Sigma and Pi Bonds Practice Problems How many sigma and pi bonds exist in a triple bond? If you said one sigma bond and two pi bonds, you are correct again! A triple bond is always made up of one sigma bond and two pi bonds. Triple bonds occur between two atoms that share six electrons (three electron pairs). Some examples of molecules with triple bonds are listed below Once a sigma bond is formed with head-to-head overlap, the only other way for two atoms to share electrons is through the side-to-side overlap of a pi bond. But why is this the case?Ī single bond is always a sigma bond and two sigma bonds cannot exist between the same atoms. If you said one sigma bond and one pi bond, you are correct! A double bond is always made up of one sigma bond and one pi bond. We are now going to explore some examples of sigma and pi bonds in the context of multiple bonds and identify how many sigma and pi bonds exist in double and triple bonds.Sigma and Pi Bonds in Double BondsSome examples of molecules with double bonds are listed belowO2 or O=ONO or N=OCO2 or O=C=Oĭouble bonds occur between two atoms that share four electrons (two electron pairs). Here is a handy table below that provides examples of each type of interaction! Type of Bond Overlapping Atomic OrbitalsĮxample Moleculessigmas-sH2, H-Hsigmap-pF2, F-F sigmahead on head s-pHCl, H-Clsigmasp2-sp2C=C in C2H4pi bondsside to side p-pO=O in O2Table 2. Pi bonds are usually formed exclusively by the side-to-side overlap of non hybridized p orbitals. Another type of interaction that creates sigma bonding is the overlap of two hybridized atomic orbitals such as sp-sp. 
The diagram above shows that sigma bonds can occur between the overlap of two s atomic orbitals, one s orbital and one p orbital or two p orbitals. Understanding these bonds can help us better understand the properties and behavior of different molecules. As you can see, different molecules have different types and numbers of sigma and pi bonds. In a molecule of acetylene (C2H2), there is one sigma bond and two pi bonds between the two carbon atoms, and one sigma bond between each carbon atom and its one hydrogen atom. In a molecule of ethene (C2H4), there is one sigma bond and one pi bond between the two carbon atoms, and one sigma bond between each carbon atom and its two hydrogen atoms. The two oxygen atoms share two pairs of electrons in two sigma bonds, and two more pairs of electrons in two pi bonds. In a molecule of O2, there are two sigma bonds and two pi bonds. The two atoms share one pair of electrons in a sigma bond. In a molecule of H2, there is only one sigma bond. Now that we know what sigma and pi bonds are, let's take a closer look at some examples in different molecules. Three types of sigma bonds between s-s, s-p, and p-p atomic orbitals and a pi bond between p-p orbitals Sigma bonds are stronger and can exist independently in single bonds, while pi bonds must coexist with a sigma bond and are only found in double and triple bonds. In summary, sigma and pi bonds are types of covalent bonds formed by different types of atomic orbital overlap. When two molecules bond, their orbitals usually combine to form hybrid orbitals like sp, sp2, and sp3. There are four types of atomic orbital sets: s, p, d, and f. Atomic orbitals are spaces where electrons are likely to be found. To understand sigma and pi bonds, you need to know a little about atomic orbitals and hybridization. 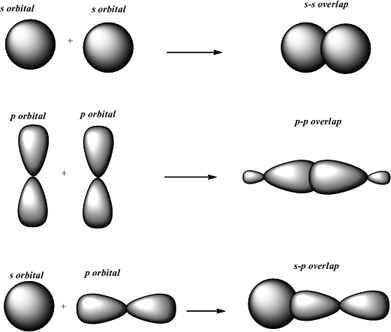
Second, sigma bonds can exist independently in single bonds, while pi bonds must coexist with a sigma bond and are only found in double and triple bonds. So, what's the difference between sigma and pi bonds? First, sigma bonds are stronger than pi bonds. They only exist in double and triple bonds. Pi bonds (π) are the second and third types of covalent bonds, formed by overlap of p orbitals side-to-side. They are found in single, double, and triple bonds. 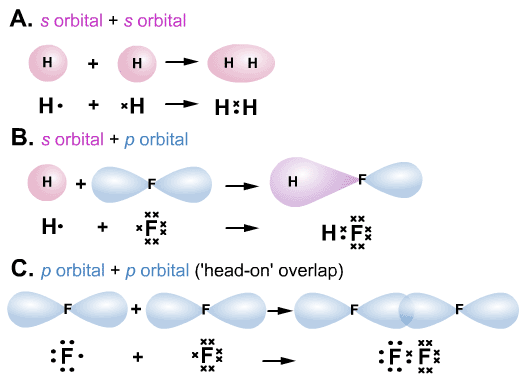
Sigma bonds (σ) are the first type of covalent bond, formed by overlap of atomic orbitals head-to-head. Covalent bonds happen when atoms share electrons. But actually, sigma and pi bonds are types of covalent bonds. When you hear the words sigma and pi bond, you might think of Greek life in college.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
